Are worms the future of drug development?
NemaMetrix’s ScreenChip collects live-animal, high-throughput C. elegans elecropharyngeograms (EPGs), which are an indication of overall organism health for drug development.
 Enlarged C. elegans. (source: National Human Genome Research Institute on Wikimedia Commons)
Enlarged C. elegans. (source: National Human Genome Research Institute on Wikimedia Commons)
At first glance, studying C. elegans feeding seems a bit esoteric. I certainly didn’t know that their swallowing frequency is an indicator of age-related health, or that monitoring this behavior is used to study neurophysiology and toxicology (Leung, 2008) for drug development. Shawn Lockery, the CTO and cofounder of NemaMetrix, compares an electropharyngeogram (EPG)—the measurement of pharyngeal pumping as electric activity—to an EKG. “[You could think of an] EKG as just the heart pushing blood around,” he said. “But you can figure out age and health and strength, all the things that are directly correlated to one of [our] most integral organs.”
Turns out, the EPG to EKG analogy is not just a literary turn of phrase. The pharyngeal pumping mechanism in C. elegans is controlled by a set of genes that determine cardiac health in humans. C. elegans possess homologues for up to 80% of human genes (Kaletta, 2006) and, as a result, are widely used as a model organism.
NemaMetrix believes that using C. elegans as an inexpensive, live-animal model can revolutionize (and democratize) the drug development process. But common methods to analyze pumping rates were archaic and cumbersome. Most often, researchers would just record a video of a worm placed on an agar plate to feed, and then by replaying in slow motion, count its swallows using a handheld counter. EPGs, pioneered by Leon Avery in 1995, record the electric activity of pharyngeal pumping. This is a more precise way to count swallow frequency and reports the action potentials of the muscles and the synaptic potentials of the neurons controlling the muscles (Figure 1-1).

But when the ScreenChip technology was first envisioned in 2008, state-of-the-art EPG recordings still relied on manual techniques and (typically) beheaded worms, making high-throughput and/or live-worm analysis impossible.
So Lockery, a University of Oregon professor studying C. elegans and electrophysiology (as well as a self-professed “maker”), imagined a device that could continuously record EGPs from a live worm. This would pave the way for a high-throughput way to measure EPGs.
The key challenge, according to Lockery, was building the right type of channel. The device would need to hold a worm securely in place between two electrodes for the EPG recording, but still allow a flow path so the worm could be exposed to solutions containing any compound of choice (Figure 1-2). At the time, Lockery was about to start a sabbatical with none other than George Whitesides, the father of microfluidics. Lockery and Whitesides lab member Elizabeth Hulme had proof of concept in just a few months. Shortly thereafter, Lockery and University of Oregon biology professor Janis Weeks cofounded NemaMetrix to commercialize the technology, bringing Matt Beaudet on board as CEO.
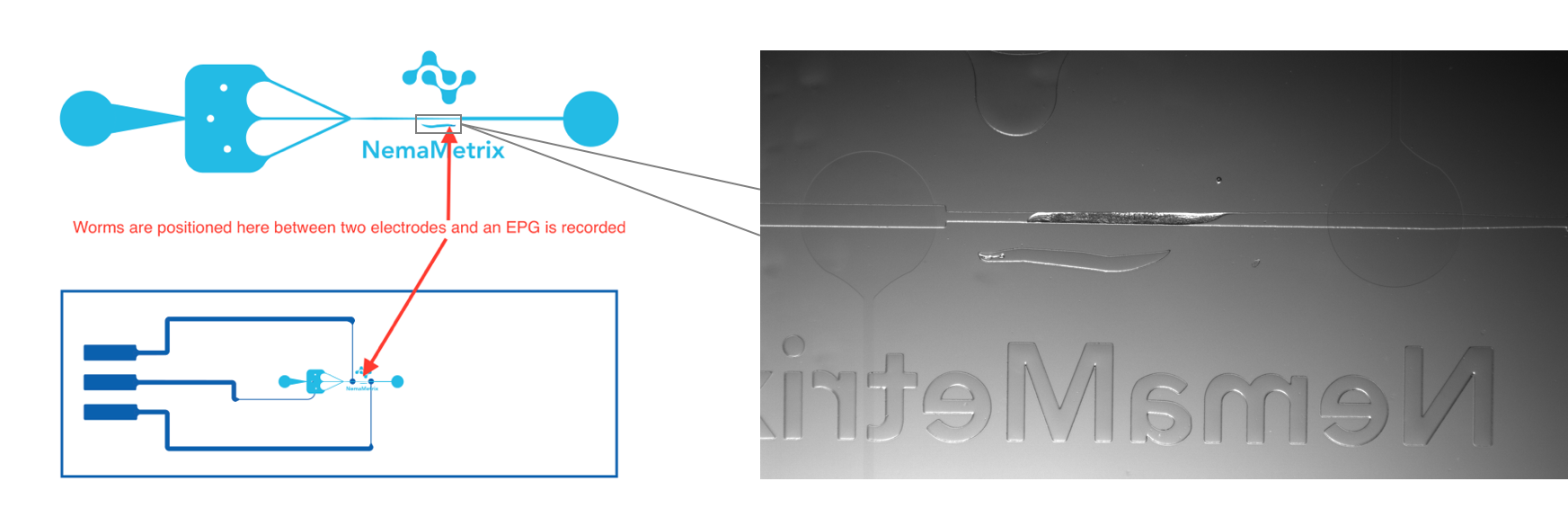
ScreenChip is designed to be used by any level of researcher and only requires a vacuum, a microscope, and a computer. Adult worms can be bulk-loaded in solutions containing any test compound, and then serially captured for the EPG recording. The system can be used for high-throughput analysis (up to 50 worms per hour) or to record EPGs on a single, live worm for as long as needed. The recorded EPGs are analyzed using open source NemAcquire software. (In fact, if you’re having trouble, the website offers to analyze your data for free!)
Though Lockery was interested in commercializing the device from the start, the ScreenChip system was initially typecast as just a handy automation tool for C. elegans researchers. Matt Beaudet thought the same thing at first glance in 2012. But then, he said, he began to “mentally overlay [the system] onto one of the big problems I’d been working on for almost a decade: How do you bridge the gap between what a cell biologist and what a mouse researcher can do?” As you progress in the typical drug development pipeline from in vitro experiments to animal models, the cost, regulatory oversight, and sheer number of experimental variables make for high stakes. Using ScreenChip, researchers can do inexpensive, quantitative live-animal work that enables them to identify leads quickly and have the rest fail fast before turning to expensive, higher-order animal models.
ScreenChip abstracts the complexity of a live model organism into a database of inputs and outputs, like in silico drug development. In Lockery’s view, “an animal is just a biological circuit.” And the motivation behind the plug-and-play design and open-source software? The founders want to demystify electrophysiology and its analysis. As Beaudet said, “This was a technology that’s the most powerful when others are innovating on top of it.”
NemaMetrix’s sights are set on pharma but are currently focused on the academic research market. Its customers are using the tool for all sorts of research, from models of cardiac arrhythmia and Alzheimer’s disease to pharmacology and toxicology. Cofounder Janis Weeks has shown that C. elegans exhibits a rapid and concentration-dependent decrease in pharyngeal pumping after exposure to the heavy metal Cu2+ (commonly generated when copper piping corrodes). Lockery has shown that nematodes get hyperphagia (better known as the munchies) after exposure to the cannabinoid anandamide, resulting in well-fed worms doubling their pumping frequency (compared to the control). NemaMetrix also has nonacademic consumers using ScreenChip to evaluate the health effects of eco-friendly detergents and public school water sources.
Though the core platform has wide applications as is, NemaMetrix wants to turn ScreenChip into a broader platform. Pharyngeal pumping is just one of many relevant C. elegans phenotypes for drug development. The company wants to build a system that can measure movement (another important indicator of animal health), as well as fluorescent phenotypes by integrating the system with a small fluorescent microscope. According to Lockery, “Modern medicine didn’t stop with EKGs—and we don’t plan to stop there either.”
