6.1. Deduction of Clausws–Clapeyron's Equations
Let AD and BC be two isothermal lines corresponding to two temperatures T and T + dT (Fig. 6.1). Let AB and CD be the liquid line and vapour line, respectively, of theAmaga’s curves. Along the border line AB, the substance is wholly in liquid state and along the line CD it is wholly in the state of saturated vapour. Let 1 gm of the substance be taken round the cycle ABCD reversibly.
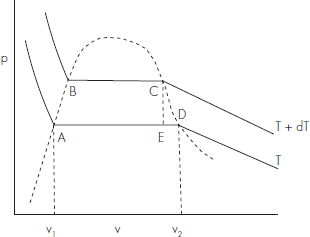
Fig. 6.1 Amagut's curves
The quantity of heat absorbed during the cycle transformation is equal to mechanical work done. We proceed to calculate the quantities of heat absorbed during the cycle process.
Heat absorbed ...
Get Heat and Thermodynamics now with the O’Reilly learning platform.
O’Reilly members experience books, live events, courses curated by job role, and more from O’Reilly and nearly 200 top publishers.

