Appendix D
Problems
Chapter 2
1. Newton had postulated that when particles collide with a surface, they lose their normal component of velocity. If this were true, and a gas in contact with a wall had equilibrium random motion of particles  , determine the relationship of wall pressure with number density (n) and temperature (T).
, determine the relationship of wall pressure with number density (n) and temperature (T).
2. Pressure is related to the flux of momentum to a wall (area). Suppose that a wall material in contact with a gas reflected the molecules with ½ of the incoming normal 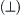 velocity component. Evaluate the energy ...
velocity component. Evaluate the energy ...
Get Introduction to Plasmas and Plasma Dynamics now with the O’Reilly learning platform.
O’Reilly members experience books, live events, courses curated by job role, and more from O’Reilly and nearly 200 top publishers.

