May 2024
Intermediate to advanced
872 pages
32h 3m
English
How to Construct Born Haber Cycle— The Easiest Way
Let us consider the Born-Haber cycle for KCl (s)
Step I Write down the equation for ΔHºf of the lattice (KCl)
![]()
Step II Write down the equation for lattice enthalpy starting from KCl (s) written above
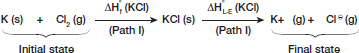
Step III Now we can construct a second pathway (path II) starting from the initial state (reactants of heat of formation) to final state (isolated gaseous ions)
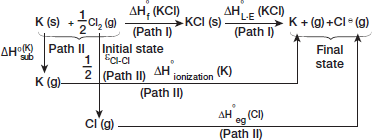
Since the net thermal change would be ...
Read now
Unlock full access