200
Methods of Non-α-Amino Acid Synthesis, Second Edition
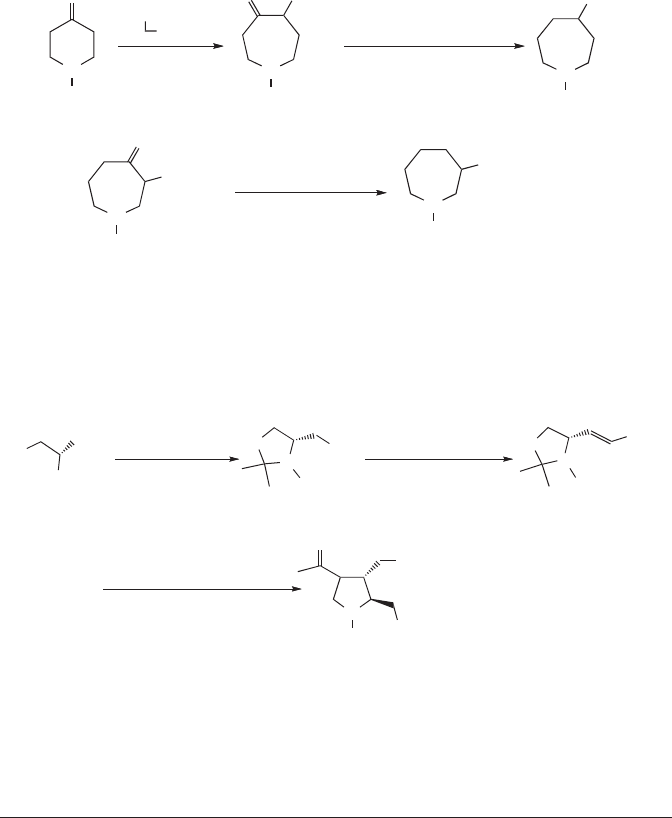
Another synthesis of heterocyclic amino acids used a ring expansion strategy. The
six-membered ring in amino-ketone 191, prepared from the N-benzyl derivative,
130
was
expanded by reaction with diazoacetate and rearrangement to give the seven-mem-
bered ring amido-ketone, 192. Reduction, elimination, and catalytic hydrogenation
gave hexahydroazepine-4-carboxylic acid, 193. The latter stages of this synthesis
were used in a closely related transformation in which a carbamoyl-protected amino-
ketone (194)
131
was converted to hexahydroazepine-3-carboxylic acid (195).
N
N
N
CO
2
Me
CO
2
Me
2
CO
